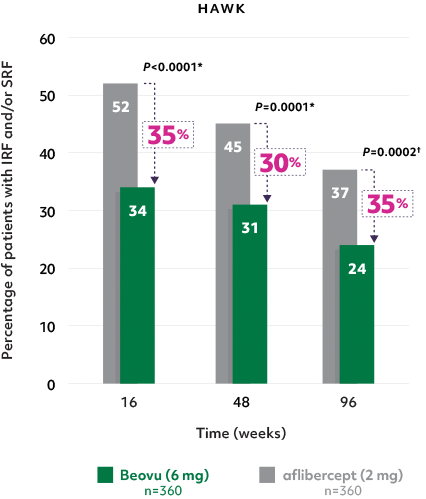
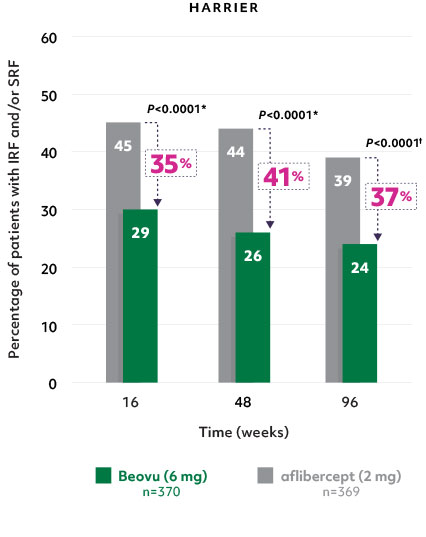
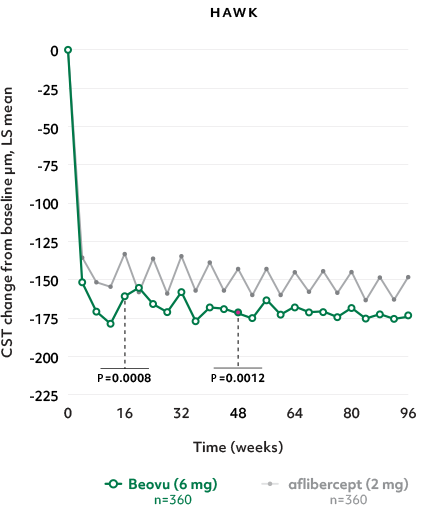
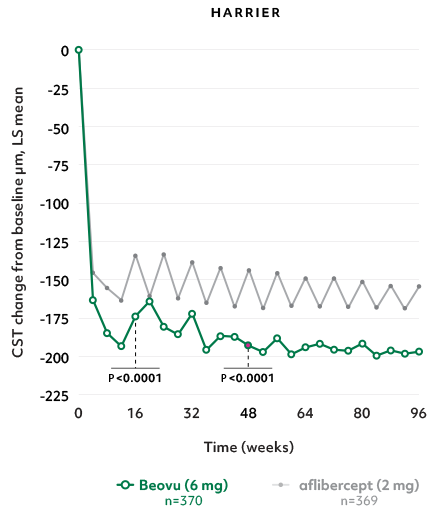
Superior fluid resolution that lasts1
• Beovu® outperformed aflibercept, with significantly fewer patients with IRF and/or SRF at Weeks 16 and 482
Patients with IRF and/or SRF on Beovu® vs aflibercept1-4
- The difference was maintained at Week 96.
Early superiority that lasts1
- Beovu® outperformed aflibercept with superior CST reductions at Weeks 16 and 481*
CST Reductions for Beovu® vs aflibercept1-4
- The difference was maintained at Week 96 (P=0.0115 in HAWK, P<0.0001 in HARRIER)4,6†‡
*Secondary endpoint in HAWK and HARRIER, confirmatory analysis in HAWK only (1-sided P values for superiority of Beovu).5
†Secondary endpoints in HAWK and HARRIER; 2-sided P values.1 ‡No hypothesis testing was performed after Week 48.
Reduce thickness. Gain letters1
- Most patients experienced vision gains and reductions in retinal thickness at Week 481
Average patient CST response3*
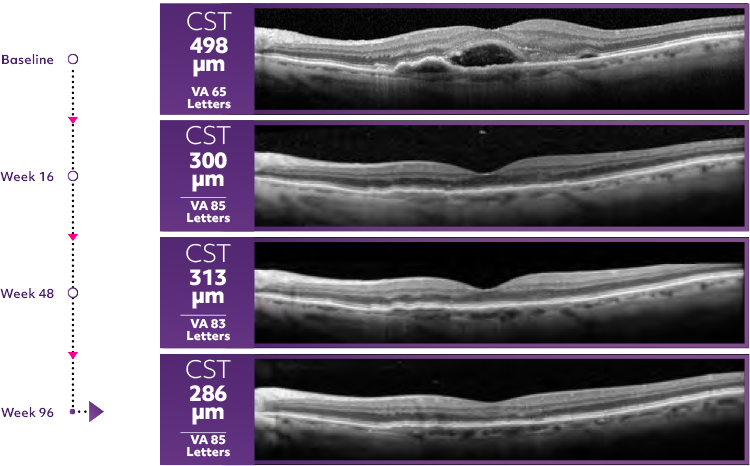
OCT scan of Beovu® patient on q12w
*OCT scans are a representation of mean patient response in HAWK.
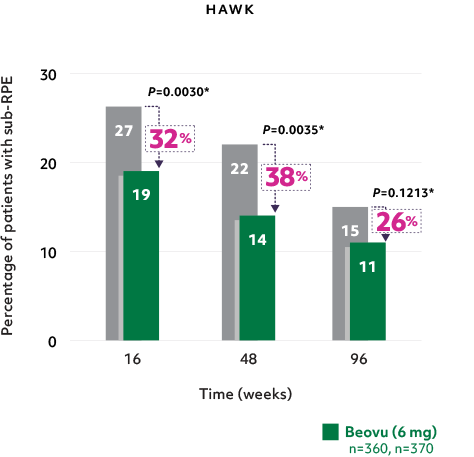
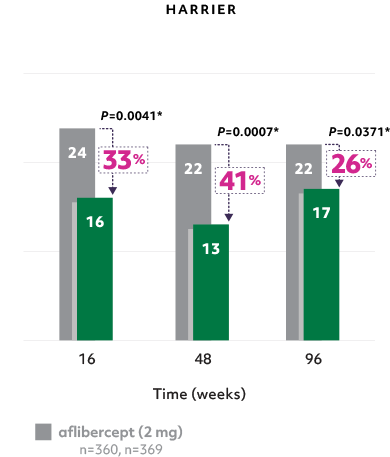
Fewer patients with sub-RPE fluid2
- Reductions in sub-RPE fluid were demonstrated at Weeks 16 and 482*
Fewer patients with sub-RPE on Beovu® vs aflibercept1-3, 5
The difference was maintained at Week 965
CST=central subfield thickness; IRF=intraretinal fluid; LS=least squares; OCT=optical coherence tomography; SRF=sub-retinal fluid
*Secondary endpoints in HAWK and HARRIER; 2-sided P values.2,3
REFERENCES
-
Dugel PU, Koh A, Ogura Y, et al; on behalf of the HAWK and HARRIER Study Investigators. HAWK and HARRIER: Phase 3, multicenter, randomized, double-masked trials of brolucizumab for neovascular age-related macular degeneration. Ophthalmology. 2020;127(1):72-123.
-
Summary of Product Characteristics. Novartis; 2020.
-
Data on file. RTH258-C002 Clinical Study Report. Novartis; 2018.
-
Data on file. RTH258-C001 & C002 IRF/SRF to 96. Novartis; 2018.
-
Data on file. RTH258-C001 Clinical Study Report. Novartis; 2018.
-
Data on file. RTH258 CST Data. Novartis; 2018.
Beovu® SmPC
Rate this content:
11/20 GLOPH/BRO/0307